${tocify}
Concepts of Research
Research refers to search for knowledge, more perfectly research is a voyage of discovery. In fact, it’s an art of careful scientific investigation. Research can be defined as the search for knowledge through objective and systematic method of finding solution to a problem. Research involves study, observation, experiments and comparison.
Background of Research /The Need for Research
Every research should have a background upon which hypothesis and research plan will be made. Background highlights the limitations of the existing status of the selected topic. It will also clarify and emphasize the fact why new investigation is required.
Purpose/Objective of Research
- To discover answers to questions through the application of scientific procedures.
- To find out the truth which is hidden and which has not yet been discovered.
- To portray (describe) accurately the characteristic of a particular group
- To determine the frequency with which something occurs
Hypothesis of Research
A hypothesis is a prediction of the outcome of the study. Hypothesis is drawn from theory and research question or from direct observations.
Hypothesis is two types:
1) Null Hypothesis and
2) Alternative Hypothesis
Example
Research Question: Do students being taught using Teaching Method A perform better at school than those being taught using Teaching Method B?
Null Hypothesis H0 : There is no difference in the final exam marks between students taught using Teaching Method A and those taught using Teaching Method B.
Alternative Hypothesis/Research Hypothesis (Hn) : There is a difference in the final exam marks between S1 students who are taught using Teaching Method A and taught using Teaching Method B.
Motivations of Research
- Desire to get a research degree along with its consequential benefits
- Desire to face the challenge in solving the unsolved problems
- Intellectual joy of doing some creative works
- Desire to serve the society and welfare for humanity
- Curiosity about new things
- Directives of government or employment conditions
Epidemeology
The Greek word epi meaning upon/among, demos meaning people and logos meaning study. Therefore Epidemeology means study upon people. Epidemiology is the study of how often diseases occur in different groups of people and why. Epidemiological information is used to plan and evaluate strategies to prevent illness and as a guide to the management of patients in whom disease has already developed.
Major areas of epidemiological study include disease etiology, transmission, outbreak investigation, disease surveillance and screening, biomonitoring, and comparisons of treatment effects such as in clinical trials.
Prevalence
Prevalence is the proportion of a population who have/had a specific characteristic (e.g. illness, depression, smoking, etc.) in a given time period. Prevalence of a disease is defined as the total number of cases of the disease in the population at a given time, or the total number of cases in the population, divided by the number of individuals in the population. It helps physicians or other health professionals understand the probability of certain diagnoses and is routinely used by epidemiologists, health care providers, government agencies and insurers.
Prevalence is usually expressed as a percentage (%), or as the number of cases per 10,000 or 100,000 people, depending on how common the illness or risk factor is in the population.
Example: the prevalence of obesity among American adults in 2001 was approximately 20.9%. This means 20.9 people among 100 was obese in America.
Incidence
Incidence is a measure of the number of new cases of a characteristic (e.g., cancer or diabetes, smoking) that arise in a population over a given period (e.g., a month, a year, etc.);
For example:
The incidence of Type 2 diabetes mellitus in Malaysia from 2013-2014 is 9% means 9% Malaysian has newly attacked by diabetes in a year (2013-2014).
Difference between Prevalence and Incidence
Prevalence is a statistical concept referring to the number of cases of a disease that are present in a particular population at a given time, whereas incidence refers to the number of new cases that develop in a given period of time.
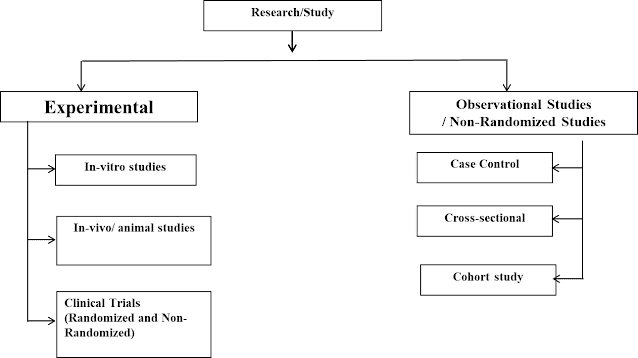
Types of Research
A. Experimental Research
i) Laboratory and Animal Studies
Experiments are done on Cells or Tissues or Animals under Strictly controlled conditions. Laboratory studies are only a starting point of investigation of therapeutic efficacy and safety or toxicological studies. In vivo studies on animals are next to in vitro studies. Both of cells and animal studies are not substitute for humans.
ii) Clinical Trials
Clinical trials are research studies on human subjects that explore whether a drug/treatment/functional foods/dietary supplement is effective and safe for humans or not. Clinical trials follow strict scientific standards which protect patients/study subjects and produce reliable study results. Clinical trials are one of the final stages of a long and careful research process. Investigators initially enroll volunteers/patients into small pilot studies, and progressively conduct large scale comparative studies.
Clinical trials are very expensive, a full series of trials may cost hundreds of millions of dollars which usually borne by the sponsor (govt. organization or pharmaceutical or biotech company). Each Clinical trial has a protocol which describes what will be done in the study, how it will be conducted, and why each part of the study is necessary. Some clinical trials need healthy volunteers while some need people with a certain disease. For safety purposes, clinical trials start with small groups of patients to find out whether a new approach or drug causes any harm.
Phases of Clinical Trials
New drug development process goes through clinical trials of 4 different phases over many years. Each phase is a separate clinical trial.
Phase I: A new drug is tested in a small group of people for the first time to evaluate its safety, determine a safe dosage range, and identify side-effects.
Phase II: The drug which passed phase I is given to a larger group of people to further evaluate its effectiveness and safety.
Phase III: The drug which passed phase II is given to large groups of people to confirm its effectiveness, monitor its side-effects, compare it to commonly ued treatments, collect information that will allow the drug to be used safely.
Phase IV: This clinical trial is conducted after the drug has been marketed to gather information on the drug’s effect in various populations and any side-effects associated with long-term use of the drug.
iii) Randomized Controlled Trial (RCT)
A Randomized Control Trial (RCT) is a type of study in which the study subjects (people) are randomly allocated either to receive or not receive the drug or to receive other treatment to compare with the drug sample.
RCT is the gold standard for a clinical trial. RCTs are used to test efficacy of drugs, safety and adverse effects of drugs, such as drug reactions.
B. Observational Studies
Observational studies involve the direct observation of patients under natural setting of treatment.
Observational study data can be two types:
Primary data:
Data for observational research is collected by the investigator for the purpose of the study. Investigator controls all aspects of the study including design, sampling techniques, data collection, and follow-up methods.
Secondary data:
The data has already been collected for another purpose but is used by the investigator to examine a novel research question. Its relatively fast and inexpensive. Sample size tend to be large. Sample may cover a large geographical area and thus provide the ability to assess national trends. But data may not include all variables of interest.
Types of Observational Studies
Observational studies may be the following types:
i) Case-Control Study
A study that compares people with a specific outcome of interest (called case; e.g., diabetes) with people from the same source population but without outcome (non-diabetic, called control). This study can be done quickly and relatively cheaply but they are not ideal for study as they gather information from the past.
ii) Cross-Sectional Study
Cross-sectional study is a type of observational study that collects information on interventions and current health outcomes from a population or group of people at one specific point in time. Information is collected from the study subjects without manipulating the study environment.
Cross-sectional study is a snapshot of a single moment in time, they don’t consider what happens before or after the snapshot is taken. It allows researchers to compare many different variables, such as, age, sex, income, education level, etc. in relation to walking and cholesterol levels at the same time, with little or no additional cost.
Examples of Cross-Sectional Study
Example-1:
Determination of cholesterol levels in daily walkers having age over 40 years and compare with non-walkers of the same age group. This investigation will not consider the past and future cholesterol levels of the study subjects.
Example-2:
Investigation of the knowledge of Pharmacy students in Malaysia about the effectiveness of herbal medicine.
iii) Cohort Study
A study in which a defined large group of people (the cohort) is closely followed over a long period of time to examine associations between different interventions received and subsequent outcomes.
Cohort studies are two types:
a) Prospective study and
b) Retrospective study
Prospective study:
This study recruits participants (cohorts/subjects/patients) before any intervention and follow them into for a long time.
Example: One might follow a cohort of middle-aged truck drivers to test the hypothesis that 20-year incidence rate of lung cancer will be highest among heavy smokers, followed by moderate smokers, and then non smokers.
Retrospective study:
This study identify subjects from past records describing the interventions received and outcomes for a certain period of time. The data obtained from the records are then analyzed for the assessment.
Example: Incidence of kidney diseases among the patients of type 2 diabetes who received treatment from University Malaya Medical Centre during 2010-2014.
iv) Longitudinal Study
Longitudinal study is an observational study which involves repeated observations of the same variables over long period of time – often many decades without interfering the study subjects.
Example: Investigation of daily walking and cholesterol levels in the body – a longitudinal study.
Steps in Conducting Research
Research activities
1. Research Based on Laboratory Experiments
In vitro, In vivo, Analysis, Formulation of Products, Preparation of Products, Development of Products, Synthesis of Products and new therapeutic biomolecules, Bioassay studies of drugs, Anti-microbial activity testing of drugs, Quality Evaluation of Drugs, etc. laboratory or experiments related works included in this category of research activities.
2. Research Works Based on Clinical Studies
This type of research involves study of drugs where human being (patients) are used as study subjects.
3. Research Based on Survey
This type of research involves a validated survey questionnaire to the people. People’s/patients’ feedback then against the questionnaire are analysed for the outcome of the study.
4. Arrangement of Scientific Seminars
Arrangement of Scientific Seminars is another important task relating to research. Scientific seminar highlights/declare the latest advancement/innovation of a specific investigation.
5. Attending in Conferences
The researchers should attend the conferences relating to their research field to know the latest advancement from other researchers all over the world as well as to present their findings to the international scientific community.
6. Publications in the journals
The researchers should publish their research achievement in a suitable peer reviewed international journal to explore their research findings to the international scientific and non-scientific communities.
Different Levels of Research
- Primary Research
The data which is collecting for the first time is called primary data and the research is called primary research. - Secondary Research
The data which is collecting from the authenticated sources including the published papers and the data are analysed for the evaluation of the impact of cumulative data is called secondary research. Examples, Systematic Review and Meta-Analysis. - Basic Research
This research is the fundamental/basic investigations. - Preliminary Research
Conduct for the preliminary evaluation of substances through screening. - Advanced/Applicable Research
The investigations which has practical applications and maintains the advancement of a specific field.
Preparations to be taken to conduct research
- Choosing a research question
- Choosing a supervisor
- Planning and developing a research project proposal
- Ethics approval
- Funding
Preparing to conduct research
1. Choosing a research question
You need to come across questions that are not answered by a review of the scientific literature. Maybe no one has done research in the area you are considering, or maybe research has been done in other countries or settings but none has been done that is applicable to your area. Once you have a research question, you need to operationalize it. This means that you need to change your question into something that is answerable by a research study.
2. Choosing a Research Supervisor:
Once you have chosen your research question, you need to find a supervisor depending on the type of research you want to do. You may find a co-supervisor to help your project.
Choose your potential supervisor who have mutual research interests. If this is your first piece of research, make sure the supervisor is willing to help you:
- Experimental and research basics
- Looking over and correcting your proposal
- Help your ethics application
- Helping you with basic questions around methodology
- Giving you feedback on your write-up the thesis
- Interested to publish your research findings in the journals or getting its patent right.
3. Developing a research project proposal
Spend some time putting together a complete study proposal with your supervisor.
This project proposal should include aims, hypotheses, methods, analysis, power calculation, timeline, information and consent forms, and proposed outcomes.
Aims should be a simple, clear explanation of the objectives.
State hypotheses based on a literature review and/or preliminary research.
Methodological detail includes: population, sampling, measurements, measurement instruments (surveys/scales/investigations).
Analysis should include detail on appropriate statistical methods, based upon statistical advice from an expert and supervisor, including the software etc. with which the statistical analysis will be conducted.
Plan a reasonable timeline for your project with your supervisor based on your commitments.
Outcomes are what you believe your research will contribute to the scientific literature and the proposed significance.
Taking the time to put together a detailed study proposal is time well-spent.
4. Ethics approval:
Ethics applications should be considered for all studies and should be discussed with your supervisor. A complete study proposal as outlined above is needed for submission for ethical approval. Information and consent forms may need to be prepared for recruitment and consent for participants. Ethics approval can be obtained from the appropriate jurisdiction:
- University Ethics Committee
- Hospital or local health service ethics committee (public or private) multiple approvals may be required in joint health service and University research
5. Funding:
Once you have put together your research proposal and received ethical approval, you can use it to apply for grants to aid you in completing the study. Your supervisor will have ideas about where to apply.
Tags:
Education